 Online calculator, figures and tables showing heat of vaporization of water, at temperatures from 0 - 370 ☌ (32 - 700 ☏) - SI and Imperial units. 
Temperatureįigures and tables showing the enthalpy and entropy of liquid water as function of temperature - SI and Imperial Units. Water - Density, Specific Weight and Thermal Expansion Coefficientsĭefinitions, online calculator and figures and tables with water properties like density, specific weight and thermal expansion coefficient of liquid water at temperatures ranging 0 to 360☌ (32 to 680☏). 
Temperature and PressureĪbsolute viscosity for water in centipoises for temperatures between 32 - 200 oF. Vicosity is a fluid's resistance to flow and can be valued as dynamic (absolute) or kinematic. Salinity - salt content - of fresh, brackish and sea water.Īttenuation of sound in seawater vs. Solubility of oxygen in equilibration with air in fresh water and seawater (salt water) - pressures ranging 1 - 4 bar abs. Oxygen - Solubility in Fresh and Sea Water vs. Viscosities of products and chemical species at varying conditions. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. Pressure and Salinityĭensities of solids, liquids and gases. Temperature and Salinityįreezing Point of Seawater vs. Temperature and SalinityĮlectrical Conductivity of Seawater vs. Salinity is normally quoted in units ‰ (parts per thousand):įor full table - rotate the screen! Temperatureĭensity of Seawater vs. Density calculated for salinity S = 35000 mg/l. air starts flowing across the surface of naphthalene with a velocity of 3 m / s. A scaling analysis based on Navier-Stokes equations is presented at the end, and the predicted θ(c) matches with experimental observations without any additional fitting parameters.Seawater temperature, saturation pressure, specific volume, specific heat, electrical conductivity and absolute viscosity. The dynamic viscosity of air at 15C is 1.78x10 5 Using Sutherlands. Furthermore, we observed a viscous wetting regime only on surfaces with an equilibrium contact angle θ(eq) smaller than a critical angle θ(c) depending on viscosity. For relatively high viscosity liquids, the inertial wetting time increases with liquid viscosity, which may due to the viscous damping of the surface capillary waves. For low viscosity liquids, the duration of inertial wetting corresponds to the time of capillary wave propagation, which can be determined by Lamb's drop oscillation model for inviscid liquids. 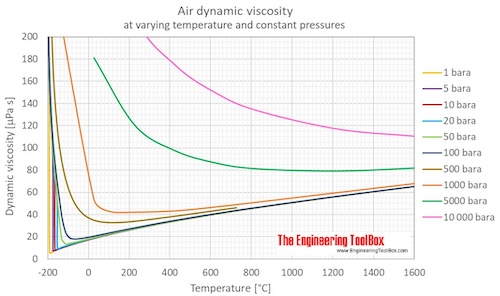
It was further found that surface wettability does not affect the duration of inertial wetting, whereas the viscosity of the liquid does. In contrast, the exponent of the power law does only depend on surface wettability as also reported in literature. In the early inertial wetting regime, the coefficient of the wetting power law increases with surface wettability but decreases with liquid viscosity. 
We show that surface wettability and liquid viscosity influence wetting dynamics and affect either the coefficient or the exponent of the power law that describes the growth of the wetting radius. Solid surfaces with different lyophilic and lyophobic coatings (equilibrium contact angle θ(eq) of 0°-112°) were used to study the effect of surface wettability. Drop of glycerol water mixtures and pure water that have comparable surface tensions (62.3-72.8 mN/m) but different viscosities (1.0-60.1 cP) were used. In this paper, we experimentally investigated the dynamic spreading of liquid drops on solid surfaces. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
